The explosion-protection methods of protection: basic concepts
ATEX device's manufacturers use different types of protection to achieve a certain EPL. These methods of protection are constantly evolving both technical and regulatory. In any case, the most used are those that have established themselves historically.
by Andrea Battauz, R&D Project Engineer of Cortem Group
Introduction
ATEX device’s manufacturers use different types of protection to achieve a certain EPL. These methods of protection are constantly evolving both technical and regulatory. In any case, the most used are those that have established themselves historically.
The in-depth knowledge of the protection methods is the prerogative of the device’s manufacturers and of the notified bodies who must verify the requirements. However, understanding some aspects of the protection modes is an essential requirement for a successful installation.
Before going into the details of the most used methods of protection, it is necessary to deepen some basic concepts which, as we will see, will return to the legislation on several occasions.
We can start by understanding the meaning of the word explosion.
Combustion and explosion
The ATEX directive defines the explosive atmosphere as: a mixture containing air, at atmospheric conditions, flammable substances in the state of gas, vapors, mists, or dusts in which, after ignition, the combustion spreads to the entire unburned mixture. [1]
The explosion covered by the ATEX directive is therefore a combustion that occurs between reactants at standard atmospheric pressure.
The combustion is an exothermic chemical reaction, which liberates heat. Normally, combustion generates heat that is released into the air without generating pressure differences. For example, a log of wood that burns in a forest or inside a fireplace, the heat has time to flow, giving thermal energy to the air and to the surrounding matter without producing sudden changes in pressure. In the case of an explosion, on the other hand, the reaction rate is such that the volume of the substances involved increases without being able to release the heat generated by the reaction, the gases expand generating pressure on the surfaces or on the gases not yet compressed.
For example: in a closed volume, starting with the reactants at atmospheric pressure, typical values of an explosion can be: 10 atmospheres of pressure peak and the 102 m/s of speed of reaction. [2]
Furthermore, compared to a common combustion reaction, the explosion is self-sustained by the propagation of a flame front through the explosive atmosphere. [3]
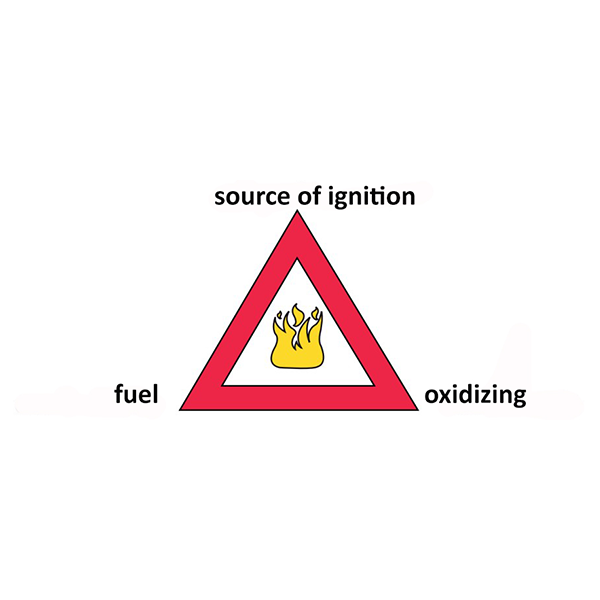
The fire triangle
To address the issue of explosion risk, as is done for the risk of fire, we talk about the schematic concept of the fire triangle.
It is a concept often reported also in fire prevention courses and in fact reflects the explosion/combustion correspondence.
The triangle summarises the three basic conditions that must be met to start a fire or explosion.
An explosion can only occur if they are present in the same place at the same time:
- Fuel
- Oxidising or oxygen
- A source of ignition
Sources of ignition in electrical equipment
Among the ignition sources, restricting the field to those most likely to be found in an electrical device, we include sparks or arcs of electrical origin, electrostatic charges, and hot surfaces (e.g., hot surface of a running engine). In addition, for specific products (such as a radio antenna or a laser instrument) electromagnetic waves, radio frequencies and optical radiation.
The mitigation of the explosion risk: basic criteria
For the design of electrical devices having an EPL Ga, Gb or Gc it will be necessary to avoid that the three factors that make up the combustion triangle are present at the same time.
To do this we will follow three basic criteria:
- Containment
- Segregation
- Prevention.
1. Containment
The explosion can occur but is confined to a well-defined volume by means of special enclosures. It is also precluded the possibility that the explosion could ignite the surrounding atmosphere. The containment of the explosion is the basis of the explosion protection method Ex-d whose enclosures are often defined flameproof.
2. Prevention
The ignition of the explosive atmosphere is avoided by controlling the energies involved, as in the intrinsically safe protection mode, or by increasing standard industrial safety criteria, as in the increased safety protection mode.This criterion is the basis of the increased safety Ex-e and intrinsically safe Ex-i protection mode
3.Segregation
The ignition sources are physically separated from the explosive atmosphere. Segregation is the basis of the protection mode by means of Ex-p pressurization, Ex-m encapsulation, Ex-o oil immersion, Ex-q powder filling, Ex- nR limited breathing.

Table 1: Basic criteria and most used protection methods
The mitigation of the risk of explosion: device failures
It is then necessary to introduce another principle which will find its declination in the various modes of protection provided for by the legislation.
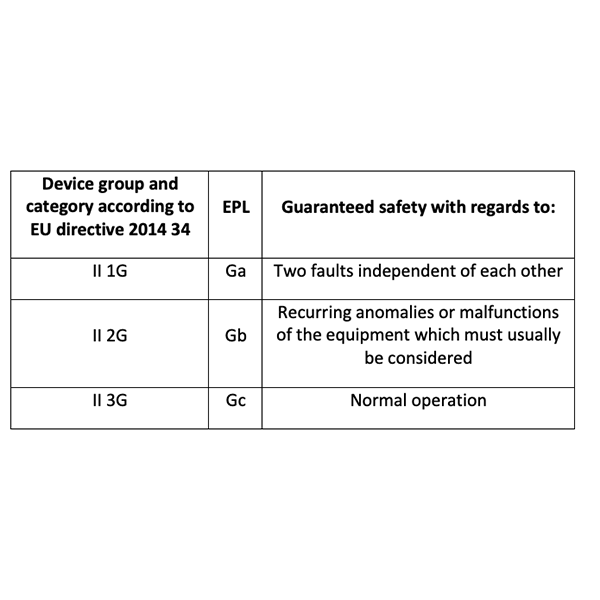
The electrical equipment that has a high EPL (protection level) must guarantee the absence of the ignition source even in fault conditions.
As shown in table 2 "Device group and category, EPL, fault tolerance [4]", this concept is also reported in the same ATEX directive.
Conclusion
In conclusion, we have seen the basic concepts underlying the main protection methods used for electrical equipment.
The choice of the protection method is linked to the type of electrical device, as well as to considerations concerning the ease of ordinary and extraordinary maintenance, the reliability of the system and, finally, the construction and maintenance costs. Notes, reference standards and bibliography
[1] DIRECTIVE 2014 34 EU - Art. 2 par 4
[2] Explosion Protection - Heinrich Groh - Elsevier - Chap. 1.1
[3] EN 1127-1: 2019 4.2.1
[4] DIRECTIVE 2014 34 EU - Annex 1 par 2